Céline Dinet, Alejandro Torres-Sánchez, Roberta Lanfranco, Lorenzo Di Michele, Marino Arroyo, Margarita Staykova
Abstract
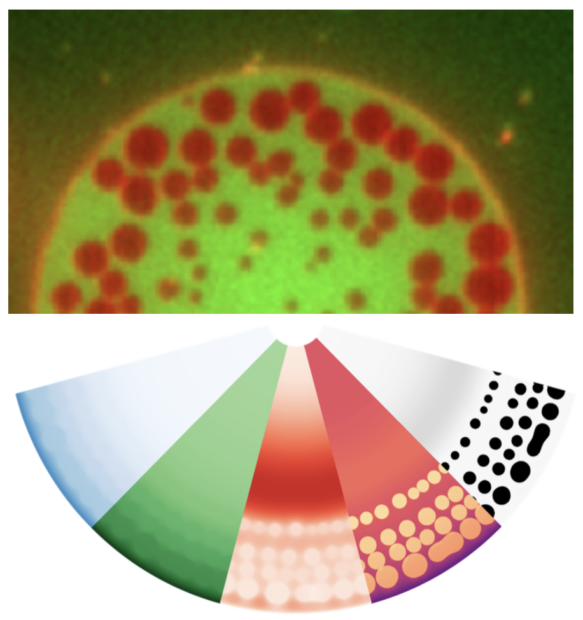
Hydraulic fracturing plays a major role in the formation of biological lumens during embryonic development, when the accumulation of pressurized fluid leads to the formation of microlumens that fracture cell-cell contacts and later evolve to form a single large lumen. However, the physical principles underpinning the formation of a pattern of microlumens from a pristine adhesion and their subsequent coarsening are poorly understood. Here, we use giant unilamellar vesicles adhered to a supported lipid bilayer and subjected to osmotic stress to generate and follow the dynamics of hydraulic fracturing akin to those in cells. Using this simplified system together with theoretical modelling and numerical simulations, we provide a mechanistic understanding of the nucleation of hydraulic cracks, their spatial patterns and their coarsening dynamics. Besides coarsening, we show that microlumens can irreversibly bud out of the membrane, reminiscent of endocytic vesicles in cell-cell adhesion. By establishing the physics of patterning and dynamics of hydraulic cracks, our work unveils the mechanical constraints for the biological regulation of hydraulically-driven adhesion remodeling.